Hydrafacial When Can I Start Hydroquinone Again
| |||
| Names | |||
|---|---|---|---|
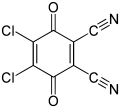
| Preferred IUPAC name 4,5-Dichloro-3,6-dioxocyclohexa-1,4-diene-i,2-dicarbonitrile[2] | |||
Other names
| |||
| Identifiers | |||
| CAS Number |
| ||
| 3D model (JSmol) |
| ||
| Abbreviations | DDQ | ||
| ChemSpider |
| ||
| ECHA InfoCard | 100.001.402 | ||
| EC Number |
| ||
| PubChem CID |
| ||
| RTECS number |
| ||
| UNII |
| ||
| CompTox Dashboard (EPA) |
| ||
| InChI
| |||
| SMILES
| |||
| Properties | |||
| Chemic formula | C 8 Cl 2 North 2 O 2 | ||
| Tooth mass | 227.00 thousand·mol−ane | ||
| Appearance | yellow to orange powder | ||
| Density | 1.7g/cm3 | ||
| Melting point | 210 to 215 °C (410 to 419 °F; 483 to 488 Yard) (decomposes) | ||
| Boiling point | 301.8 °C (575.2 °F; 575.0 K) at 760mmHg | ||
| Solubility in water | reacts | ||
| Hazards | |||
| GHS labelling: | |||
| Pictograms |  | ||
| Signal word | Danger | ||
| Hazard statements | H301 | ||
| Precautionary statements | P264, P270, P301+P310, P321, P330, P405, P501 | ||
| Flash indicate | 136.3 °C (277.3 °F; 409.iv G) | ||
| Except where otherwise noted, information are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| | |||
| Infobox references | |||
ii,3-Dichloro-v,6-dicyano-i,iv-benzoquinone (or DDQ) is the chemical reagent with formula C6Cl2(CN)2O2. This oxidant is useful for the dehydrogenation of alcohols,[3] phenols,[4] and steroid ketones.[5] DDQ decomposes in h2o, but is stable in aqueous mineral acid.[six]
Training [edit]
Synthesis of DDQ involves cyanation of chloranil. J. Thiele and F. Günther starting time reported a 6-pace training in 1906.[7] The substance did not receive interest until its potential as a dehydrogenation amanuensis was discovered. A single-pace chlorination from two,3-dicyanohydroquinone was reported in 1965.[8]
Reactions [edit]
The reagent removes pairs of H atoms from organic molecules. The stoichiometry of its action is illustrated by the conversion of tetralin to naphthalene:
- 2 C6Cl2(CN)iiO2 + C10H12 → 2 C6Cl2(CN)2(OH)two + C10H8
The resulting hydroquinone is poorly soluble in typical reaction solvents (dioxane, benzene, alkanes), which facilitates workup.
Solutions of DDQ in benzene are red, due to the formation of a charge-transfer complex.[9]
Dehydrogenation [edit]
Aromatization [edit]
-
 [10]
[10]
Cross-Dehydrogenative Coupling [edit]
-

- [11]
Safety [edit]
DDQ reacts with h2o to release highly toxic hydrogen cyanide (HCN). A depression-temperature and weakly acidic environment increases the stability of DDQ.
References [edit]
- ^ 2,3-Dichloro-five,6-dicyano-p-benzoquinone at Sigma-Aldrich
- ^ Classification of Organic Chemistry : IUPAC Recommendations and Preferred Names 2013 (Blueish Book). Cambridge: The Purple Guild of Chemistry. 2014. p. 50. doi:ten.1039/9781849733069-FP001. ISBN978-0-85404-182-iv.
- ^ Braude, E. A.; Linstead, R. P.; Wooldridge, K. R. H. (1956). "593. Hydrogen Transfer. Function 9 The Selective Dehydrogenation of Unsaturated Alcohols by High-potential Quinones". Journal of the Chemical Society (Resumed). 1956: 3070–3074. doi:10.1039/JR9560003070.
- ^ Becker, H. D. (1965). "Quinone Dehydrogenation. I. Oxidation of Monohydric Phenols". Journal of Organic Chemistry. 30 (4): 982–989. doi:10.1021/jo01015a006.
- ^ Turner, A. B.; Ringold, H. J. (1967). "Applications of High-potential Quinones. Part I. The Machinery of Dehydrogenation of Steroidal Ketones by 2,3-Dichloro-5,half-dozen-Dicyanobenzoquinone". Journal of the Chemical Society C: Organic. 1967: 1720–1730. doi:10.1039/J39670001720.
- ^ Derek R. Buckle, Steven J. Collier, Marking D. McLaws "2,3-Dichloro-v,6-dicyano-1,four-benzoquinone" in E-EROS ENCYCLOPEDIA OF REAGENTS FOR ORGANIC SYNTHESIS, 2005. doi:10.1002/047084289X.rd114.pub2
- ^ Thiele, J.; Günther, F. (1906). "Ueber Abkömmlinge des Dicyanhydrochinons". Justus Liebig'due south Annalen der Chemie. 349 (one): 45–66. doi:x.1002/jlac.19063490103.
- ^ Walker, D.; Waugh, T. D. (1965). "2,3-Dichloro-5,6-Dicyanobenzoquinone (DDQ). A New Training". The Journal of Organic Chemical science. 30 (9): 3240. doi:ten.1021/jo01020a529.
- ^ Rathore, Rajendra; Kochi, Jay K. (2000), "Donor/acceptor organizations and the electron-transfer image for organic reactivity", Advances in Physical Organic Chemical science, Elsevier, pp. 193–318, doi:ten.1016/s0065-3160(00)35014-vi, ISBN9780120335350
- ^ Brown, Westward.; Turner, A. B. (1971). "Application of High-potential Quinones. vii. Synthesis of Steroidal Phenanthrenes by Double Methyl Migration". Journal of the Chemical Guild C: Organic. 1971: 2566–2572. doi:10.1039/J39710002566. PMID 5167256.
- ^ Zhang, Y.; Li, C. J. (2006). "DDQ-Mediated Directly Cross-Dehydrogenative-Coupling (CDC) between Benzyl Ethers and Unproblematic Ketones". Journal of the American Chemical Society. 128 (thirteen): 4242–4243. doi:x.1021/ja060050p. PMID 16568995.
External links [edit]
- "Like Neurons in the Brain": A Molecular Figurer that Evolves
Source: https://en.wikipedia.org/wiki/2,3-Dichloro-5,6-dicyano-1,4-benzoquinone


0 Response to "Hydrafacial When Can I Start Hydroquinone Again"
Post a Comment